Demographic Shift Means Lower Birthrates and Aging Populations around the World, Suggesting Big Changes for Global Healthcare, Pathology Groups, and Clinical Laboratories
Demographic shifts are most acute in Europe and East Asia but could be a harbinger of things to come for US healthcare as well
Across the globe, major shifts in many countries’ demographics are starting to drive notable changes in how healthcare is delivered in these nations. Having fewer pediatric patients and more senior citizens is fundamentally altering what types of tests are in greatest demand from the medical laboratories in these countries. It is the population trend writ large on a global scale.
For example, in countries as diverse as Sweden, Hungary, Japan, and South Korea, birthrates are declining as fewer young people decide to have kids, or they choose to have smaller families. Thus, demand for pediatric care is declining in those countries.
Meanwhile, populations around the world continue to age as greater numbers of people reach their retirement years. Not only does this create the need to expand medical services designed to serve the elderly, but there are important economic consequences. That’s because each wave of retirees leaves fewer people in the workforce to support the healthcare of ever-growing numbers of senior citizens.
According to The New York Times (NYT), this trend is likely to accelerate. In “Long Slide Looms for World Population, with Sweeping Ramifications,” the paper reported that “All over the world, countries are confronting population stagnation and a fertility bust, a dizzying reversal unmatched in recorded history that will make first-birthday parties a rarer sight than funerals, and empty homes a common eyesore.”
The NYT added that, “With fewer births, fewer girls grow up to have children, and if they have smaller families than their parents did—which is happening in dozens of countries—the drop starts to look like a rock thrown off a cliff.”
In countries such as the US, Canada, and Australia, this is partially mitigated by immigration, the NYT reports. However, some nations, such as Germany and South Korea, have instituted programs aimed at boosting birthrates, though with varying degrees of success.
According to demographer Frank Swiaczny, Dr. rer. nat., Senior Research Fellow at the Federal Institute for Population Research in Germany, countries around the world—especially in Europe and East Asia—“need to learn to live with and adapt to decline.”
“A paradigm shift is necessary,” he told the NYT.
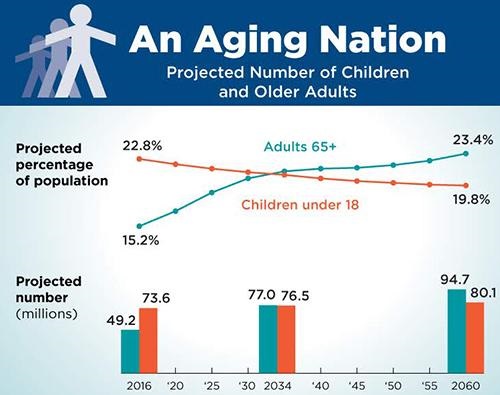
The graphic above, taken from the US Census Bureau’s 2018 report, “The Graying of America: More Older Adults than Kids by 2035,” illustrates the rate at which America’s elder population is catching up with the rest of the world. It will soon exceed younger portions of the population, thus shifting demand for healthcare from pediatrics to geriatrics. Anatomic pathology groups and clinical laboratories will be impacted by this trend. (Graphic copyright: US Census Bureau.)
Elder Population Growth: Academics Take Notice
Healthcare scholars also have been looking at the topic of demographic shift. A recent commentary in Health Affairs, titled “Actualizing Better Health and Health Care for Older Adults,” focused on the policy implications for senior care.
The authors, which included Terry Fulmer PhD, RN, FAAN, and John Auerbach, Director of Intergovernmental and Strategic Affairs at the CDC, noted that in 2018, adults 65 or older were 15.6% of the population. This will rise to 20% by 2030, when, according to the authors, seniors will outnumber the portion of the population that is younger than age five.
Fulmer is President of the John A. Hartford Foundation, which is dedicated to improving care for older adults, and until May, Auerbach was President and CEO of Trust for America’s Health (TFAH).
They recommended six broad policy goals:
- Foster an “expanded and better-trained workforce” to care for older adults, through enhanced training as well as “scholarships, loan forgiveness, and clinical internships.”
- Adapt the public health system to account more for the needs of an aging population, such as by “improved coordination and collaboration with Area Agencies on Aging and key healthcare providers.”
- Address disparities and inequities in healthcare access, such as social isolation “caused or exacerbated by social, economic, and environmental conditions.”
- Facilitate advances in telehealth and other technologies to improve care delivery. “The lack of access to technology, low digital health literacy, and design barriers in patient portals and apps have disproportionately affected older adults, especially those in underserved communities,” the authors wrote.
- Improve palliative and end-of-life care. “Many older adults are living with serious illness,” the authors wrote, and “most will live for years with their illnesses, resulting in a high burden of physical and psychological distress, functional dependence, poor quality of life, high acute care use, loss of savings, and caregiver distress.”
- Reform long-term care, by improving conditions in long-term care facilities and making it easier for older adults to stay at home.
The authors also urged a move away from “traditional fee-for-service Medicare” through “policy changes such as bundled, capitated, and other value-based payments.”
A perspective in the journal NPJ Urban Sustainability, titled “Ageing and Population Shrinking: Implications for Sustainability in the Urban Century,” notes that these trends have led some cities or countries to adopt technological innovations in healthcare, such as “socially assistive robots and virtual entertainment for mental health, roadside AI services for healthcare, and a series of innovations for house-based healthcare, digital nursing, and monitoring.”
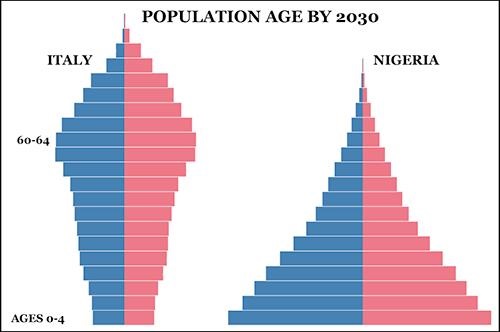
The graphic above, taken from PopulationPyramid.net, illustrates the stark differences in the age of populations in two countries at opposite ends of the progressing demographic shift. Italy’s population pyramid (left) shows how the senior population makes up a substantial proportion of total population, while Nigeria’s 2030 population pyramid (right) shows the classic pyramid of a wide base of younger people trailing off to a small number of the elderly at the top of the pyramid. Medical laboratories in those nations will continue to be affected by how these demographic shifts taking place worldwide are changing the type of healthcare in highest demand. (Graphic copyright: PopulationPyramid.net.)
Impact on Pediatrics
At the other end of the age spectrum, a recent presentation from the American Academy of Pediatrics noted a 13% decline in the US birthrate between 2007 and 2019. But a white paper from physician search firm Merritt Hawkins suggests this has not necessarily resulted in reduced demand for pediatric services, at least not in the US.
Despite the decline, “there are still about four million births in the US annually, and immigration adds to the number of children in the population,” the white paper notes. Even rural areas with aging populations “have far fewer pediatricians per capita than they require.”
However, according to The New York Times, in South Korea, “expectant mothers in many areas can no longer find obstetricians or postnatal care centers.” And the town of Agnone, Italy, no longer has a maternity ward because the number of births—just six this year—is below the national minimum.
This is important to note. If there are developed countries around the world where demographics point to a steady decline in population, then the type of healthcare provided will be different than what is currently used. Clinical laboratories and pathology groups in those regions can expect changes and should prepare for them.
—Stephen Beale
Related Information:
Long Slide Looms for World Population, with Sweeping Ramifications
Aging and Population Shrinking: Implications for Sustainability in the Urban Century




