Apr 9, 2018 | Laboratory Pathology
Direct-to-consumer (DTC) genetic testing has been much in the news of late and clinical laboratories, anatomic pathology groups, and biomedical researchers have a stake in how the controversy plays out.
While healthcare consumers seem enamored with the idea of investigating their genomic ancestry in growing numbers, the question of how the data is collected, secured, and distributed when and to whom, is under increased scrutiny by federal lawmakers, bioethicists, and research scientists.
However, should public demand for DTC testing find support in Congress, some lab companies offering direct-to-consumer genetic tests could find their primary source of revenue curtailed.
DTC Sales Skyrocket as FDA Authorizes Genetic Tests for Certain Chronic Diseases
Dark Daily reported last fall on one company that had its plans to distribute thousands of free genetic tests at a football game suspended due to privacy concerns. (See, “State and Federal Agencies Throw Yellow Flag Delaying Free Genetic Tests at NFL Games in Baltimore—Are Clinical Laboratories on Notice about Free Testing?” October 13, 2017.)
Nevertheless, consumer demand for DTC tests continues to rise. In a press release, Ancestry, a family genetic history and consumer genomics company, reported:
- Record sales of AncestryDNA kits during the 2017 four-day Black Friday to Cyber Monday weekend, selling more than 1.5 million kits; and,
- The 2017 sales were triple the amount of kits sold during the same period in 2016.
Possibly helping the sale of DTC genetic tests may be the US Food and Drug Administration (FDA) authorization last year of 23andMe’s Personal Genome Service Genetic Health Risk tests for 10 diseases or conditions, including:
Senator Calls for Investigation of DTC Genetic Test Company Use of Patient Data
These are impressive sales. However, medical professionals may wonder how so much genetic data can be kept private by the testing companies. And medical laboratory leaders are not the only ones asking about privacy and the use of genetic test results.
In a November press conference, Senate Minority Leader Chuck Schumer called on the Federal Trade Commission (FTC) to look into genetic testing companies’ privacy and disclosure practices, noted NBC News.
“What those companies can do with all that data—your most sensitive and deepest info, your genetics—is not clear, and in some cases not fair and not right,” stated Schumer.
Congress took action in 2008 by passing the Genetic Information and Nondiscrimination Act (GINA), which bans employers and insurers from making decisions about people based on genetic predispositions to disease.
However, lawmakers also recently introduced House Bill 1313, the Preserving Employee Wellness Programs Act. It reads, in part, “… the collection of information about the manifested disease or disorder of a family member shall not be considered an unlawful acquisition of genetic information with respect to another family as part of a workplace wellness program offered by an employer ….”
“We’re injecting terrible opportunities for discrimination in the workplace,” Robert Green, MD, Professor of Medicine (Genetics) at Harvard Medical School, told Gizmodo.

Robert C. Green, MD, MPH (above), Professor of Medicine, Harvard Medical School; Associate Physician, Brigham and Women’s Hospital; Geneticist, Brigham and Women’s Hospital; and Director, Genomes2People Research Program at Brigham and Women’s Hospital, believes weak genetic privacy laws are inhibiting research and clinical care. “People decline genetic tests because of concerns over privacy and genetic discrimination, especially insurance discrimination,” he told Gizmodo. “This is stymying biomedical research and people’s access to healthcare.” (Photo copyright: Harvard Medical School.)
HIPAA Enables Selling of Anonymized Patient Genetic Data
Peter Pitts, former FDA Associate Commissioner, and President and Co-founder of the Center for Medicine in the Public Interest, a non-profit medical issues research group, blames the release of data by DTC genetic test companies on the Health Insurance Portability and Accountability Act (HIPAA), a law he says makes way for “anonymized” sale of patient data.
“The Portability Act was passed when genetic testing was just a distant dream on the horizon of personalized medicine,” Pitts wrote in a Forbes commentary. “But today that loophole has proven to be a cash cow. 23andMe has sold access to its database to at least 13 outside pharmaceutical firms … AncestryDNA recently announced a lucrative data-sharing partnership with the biotech company Calico.”
For its part, in an online privacy statement, 23andMe noted, “We will use your genetic information or self-reported information and share it with third parties for scientific research purposes only if you sign the appropriate consent document.”
Similarly, Ancestry points out in its posted privacy statement, “We share your genetic information with research partners only when you provide us with your express consent to do so through our informed consent to research.
Consumers Speak Out on Privacy; States Study Laws and Genetic Testing by Research Hospitals
How do consumers feel about the privacy of their genetic test data? According to a news release, a survey by 23andMe found the following:
- 80% of Americans are concerned about DNA testing privacy; however,
- 88% have no awareness or understanding of what testing companies do to protect information; and,
- 74% of people are, nonetheless, interested in genetic testing.
Meanwhile, as states promulgate various genetic privacy laws, a paper published at SSRN by researchers at the Massachusetts Institute of Technology (MIT) and the University of Virginia (UV) examined how different state laws affect patients’ decisions about having genetic testing performed at various research hospitals.
The MIT/UV study focused on genetic testing by research hospitals as opposed to the DTC genetic testing by private companies. The paper explained that states have one of three types of laws to protect patients’ privacy in genetic testing:
- “Require the provider to notify the individual about potential privacy risks;
- “Restrict discriminatory use of genetic data by employers or insurance companies; and,
- “Limit redisclosure without consent.”
Findings, netted from more than 81,000 respondents, suggest:
- When genetic data are explained in state laws as patient property, more tests are performed;
- Conversely, state laws that focus on risk, and ask patients to consent to risk, lead to less people giving the go-ahead for genetic testing.
“We found a positive effect [on the number of tests] was an approach where you gave patients the potential to actually control their own data,” Catherine Tucker, PhD, Distinguished Professor of Management at MIT and one of the study researchers, told MIT News.
Whether the provider of genetic tests is a private testing company or a research hospital’s clinical laboratory, privacy continues to be a concern, not just to physicians but to federal lawmakers as well. Nevertheless, healthcare consumers and patients who receive comprehensible information about how their genetic data may be used seem to be agreeable to it. At least for now, that is.
—Donna Marie Pocius
Related Information:
AncestryDNA Breaks Holiday Sales Record Black Friday to Cyber Monday
Senator Calls for More Scrutiny of Home DNA Test Industry
The Present and Future Asymmetry of Consumer Genetic Testing
Are Our Terrible Genetic Privacy Laws Hurting Science?
The Privacy Delusions of Genetic Testing
National Survey Shows Strong Interest in DNA Testing
Privacy Protection, Personalized Medicine, and Genetic Testing
How Privacy Policies Affect Genetic Testing
State and Federal Agencies Throw Yellow Flag Delaying Free Genetic Tests at NFL Games in Baltimore—Are Clinical Laboratories on Notice about Free Testing?
Jan 26, 2018 | Instruments & Equipment, Laboratory Instruments & Laboratory Equipment, Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Sales and Marketing, Laboratory Testing, Management & Operations
Lack of Medicare or third-party payer coverage for most genetic screening tests in healthy adults is not discouraging development of new gene testing products
With the global anatomic pathology genetic testing market poised to reach $9.8 billion by 2025, clinical laboratories continue to develop new genetic screening tests (rather than diagnostic tests) intended to help physicians identify patients who carry inherited genetic mutations that could put them or their future children at higher risk for chronic disease, such as cancer.
This is a bit of a gamble since (with some exceptions) Medicare and many health insurers typically will not pay for predictive and presymptomatic genetic tests and services used to detect an undiagnosed disease or disease predisposition.
Nevertheless, Inkwood Research of Gurugram, India, predicts in its “Global Genetic Testing Market Forecast 2017-2024” report that aging populations throughout the world will be the driving force producing “enormous opportunities for the global genetic testing market.” The research firm anticipates this will result in a 9.93% increase in annual sales revenue during each of the next seven years.
Screening versus Diagnostic Testing Gains Popularity Among Patients, Physicians
Genetic diagnostic testing promises to accelerate the growth of precision medicine by guiding the diagnosis and treatment of cancer and other chronic diseases. However, genetic tests that “screen” healthy patients for predispositions to certain diseases also are gaining traction in the marketplace.
The US Food and Drug Administration (FDA) gave direct-to-consumer genetic screening testing a boost in April 2017 when it allowed marketing of 23andMe Personal Genome Service Genetic Health Risk tests for 10 inherited diseases or conditions, including:
· Parkinson’s Disease;
· Late-onset Alzheimer’s Disease;
· Celiac Disease; and
· other conditions.
“Consumers can now have direct access to certain genetic risk information,” Jeffrey Shuren, MD, Director of the FDA’s Center for Devices and Radiological Health, said in a press release. “But it is important that people understand that genetic risk is just one piece of the bigger puzzle, it does not mean they will or won’t ultimately develop a disease.”
Robert Green, MD, MPH, a Professor of Medicine at Harvard Medical School, told NPR that consumers should have access to genetic information. However, they also need to understand its limitations.
“Some people really want this [genetic] information on their own, and others want it through their physician,” Green said. “Both those channels are legitimate. People should just be aware that this information is complicated.”
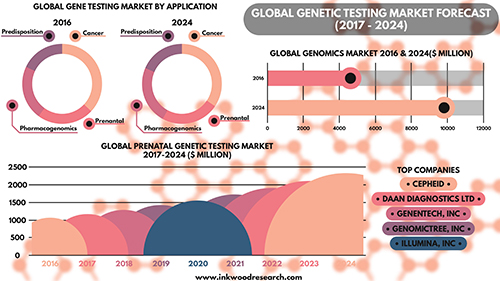
According to the Inkwood Research report, “The global genetic testing market is anticipated to grow from $4,614 million in 2016 to $9,806 million by 2025, at a CAGR [Compound Annual Growth Rate] of 9.93% between 2017 and 2025. The important driver increasing growth in the global genetic testing market is an aging population on the rise. The rising geriatric population is driving the global genetic testing market to a significant level.” (Caption and graphic copyright: Inkwood Research.)
· Cystic Fibrosis;
· Sickle Cell Disease; and
· Spinal Muscular Atrophy.
The genetic screening panel tests for the 22 heritable diseases cited by the American College of Obstetricians and Gynecologists (ACOG) in a Committee Opinion on genetic carrier screenings published by the ACOG in March 2017.
“The United States is truly a melting pot, and it no longer makes sense for physicians to assume genetic screening is appropriate for an individual based on presumed race or ethnicity,” Felicitas Lacbawan, MD, Executive Medical Director, Advanced Diagnostics, Quest Diagnostics, stated in a press release. “QHerit is designed for any woman and her partner, not just those in a specific, so-called high-risk ethnic or racial group.”
Genetic Screening in Primary Care Helps Assess Risk for Chronic Disease
Genetic diagnostic test developer Invitae (NYSE:NVTA) also points to growing evidence of the genetic screening test’s value to healthy individuals. In September 2017, Invitae presented initial findings at the National Society of Genetic Counselors 36th Annual Conference. The study showed a retrospective analysis of 120 patients tested with a proactive genetic screening panel for healthy adults had revealed medically significant findings for nearly one in five patients.
“Interest among otherwise healthy adults in using genetic information to understand their risk of disease conditions continues to grow each year, ” Robert Nussbaum, MD, Chief Medical Officer of Invitae, said in a press release. “These and other data show that interest is well-placed, with a substantial group of patients showing genetic variants associated with elevated risk of diseases like cancer where monitoring and early intervention can be helpful. Use of genetic screening in the primary care setting can assess risk to help shape individual screening plans. We are continually adding tools and resources that help reduce barriers to the widespread use of genetic information in mainstream medical practice.”
Routine Genetic Screening Could Become Norm, CDC Says
The Centers for Disease Control and Prevention (CDC) notes that newborn screening is “currently the largest public health genetics program in the world,” with more than four million babies screened at birth each year for 30 or more genetic conditions. In the CDC’s “Genomics and Health Impact Blog,” the agency continues to maintain a “cautionary attitude about personal genomic tests” beyond the newborn period, directing those considering direct-to-consumer laboratory testing, such as 23andMe and MyMedLab, to “think before you spit.”
Nonetheless, the CDC acknowledges routine genetic screening of healthy people could become the norm. However, others advise caution.
“To be sure, while the use of genome sequencing is promising in certain clinical scenarios, such as rare diseases and cancer, we do not think that whole genome sequencing in the general population is appropriate at this time,” Muin J. Khoury, PhD, MD, Director, Office of Public Health Genomics, CDC, wrote in a January 30, 2017, blog post. “We would not recommend its use outside research studies … But it is also becoming clearer that as science progresses, we are discovering more opportunities for using genetic screening of healthy individuals for preventing common diseases across the lifespan, outside of the newborn screening context.”
The impact on clinical laboratories and anatomic pathology groups should genetic screening become normalized should be clear: Labs will be tasked with performing these tests, and pathologists will be needed to interpret them and educate both physicians and patients on the findings.
Before that, however, genetic screening tests will need to be fully supported by government, and insurers, including Medicare, will have to agree to pay for them.
—Andrea Downing Peck
Related Information:
Global Genetic Testing Market Forecast 2017-2024
Carrier Screening for Genetic Conditions
Quest Diagnostics Launches QHerit, a Pan-Ethnic Genetic Screening Panel Aligned with New Medical Guidelines
Invitae Expands Test Menu for Proactive Genetic Testing in Healthy Adults
Invitae Highlighting New Research, Expanded Suite of Services at National Society of Genetic Counselors (NSGC) 36th Annual Conference
Consumer Genetic Testing: Think Before You Spit, 2017 Edition
Genetic Screening of Healthy Populations to Save Lives and Prevent Disease
FDA Allows Marketing of First Direct-to-Consumer Test that Provide Genetic Risk Information for Certain Conditions
FDA Approves Marketing of Consumer Genetic Tests for Some Conditions





