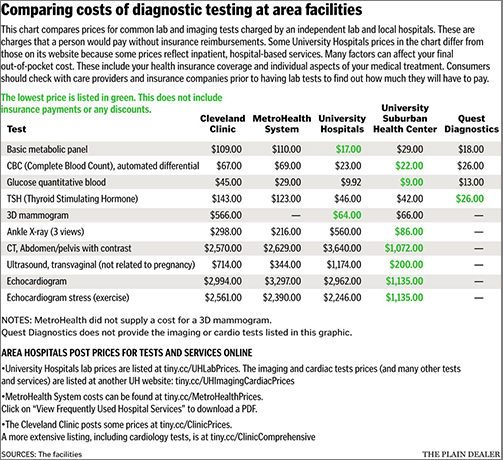
Multiple Pathology and Other Healthcare Organizations Request CDC Include Clinical Laboratory Personnel in First Round of COVID-19 Vaccinations
CAP president maintains medical laboratory staff are ‘indispensable’ in pandemic fight and should be in ‘top tier’ for vaccination
As COVID-19 vaccinations continue to roll out, the College of American Pathologists (CAP) is lobbying for clinical pathologists and medical laboratory staff to be moved up the priority list for vaccinations, stating they are “indispensable” in the pandemic fight.
In a news release, CAP’s President Patrick Godbey, MD, FCAP argued for the early vaccination of laboratory workers, “It is essential that early access to the vaccine be provided to all pathologists and laboratory personnel,” he said. “Pathologists have led throughout this pandemic by bringing tests for the coronavirus online in communities across the country and we must ensure that patient access to testing continues. We must also serve as a resource to discuss the facts about the vaccine and answer questions patients, family members, and friends have about why they should get the vaccine when it is available to them.”
Who Does CDC Think Should Be First to Be Vaccinated?
According toThe New York Times (NYT), there are an estimated 21 million healthcare workers in the United States, making it basically “impossible,” the NYT wrote, for them all to get vaccinated in the first wave of COVID-19 vaccinations.
A December 11, 2020, CDC Morbidity and Mortality Weekly Report, titled, “ACIP Interim Recommendation for Allocating Initial Supplies of COVID-19 Vaccine—United States, 2020,” notes that “The [federal] Advisory Committee on Immunization Practices (ACIP) recommended, as interim guidance, that both 1) healthcare personnel and 2) residents of long-term care facilities be offered COVID-19 vaccine in the initial phase of the vaccination program.”
The ACIP report defines healthcare personnel as “paid and unpaid persons serving in healthcare settings who have the potential for direct or indirect exposure to patients or infectious materials.”
However, a CDC terminology guidance document listed at the bottom of the ACIP report states, “For this update, HCP [Healthcare Personnel] does not include dental healthcare personnel, autopsy personnel, and laboratory personnel, as recommendations to address occupational infection prevention and control (IPC) services for these personnel are posted elsewhere.”
On December 16, the American Society for Clinical Pathology (ASCP) called attention to this discrepancy by sending a letter to CDC Director Robert R. Redfield, MD. The letter was co-signed by the:
- American Association for Clinical Chemistry (AACC),
- American Medical Technologists (AMT),
- American Society for Clinical Laboratory Science (ASCLS),
- American Society for Clinical Pathology (ASCP),
- American Society of Cytopathology (ASC),
- Association of Pathology Chairs (APC),
- National Independent Laboratory Association (NILA),
- As well as 18 other healthcare organizations.
In part, the letter stated, “We are convinced that ACIP did not intend to exclude any healthcare workers from its recommendation to offer vaccinations to healthcare personnel in the initial phase of the COVID-19 vaccination program (Phase 1a). However, we would hate for jurisdictions to overlook dental, autopsy, and laboratory personnel because of a minor footnote in [CDC] guidance that was developed for an entirely different purpose (i.e., infection control).
“We respectfully ask CDC to clarify,” the letter continues, “… that all healthcare workers—including dental, autopsy, and laboratory personnel—are among those who should be given priority access to vaccine during the initial phase of the COVID-19 vaccination program.”
Forgotten Frontline Healthcare Workers?
Clinical laboratory professionals continue to maintain they should be in the first priority grouping, because they are in direct contact with the virus even if they are not directly interacting with patients. In the CAP virtual press conference streamed on Dec. 9, 2020, Godbey; Amy Karger, MD, PhD, faculty investigator at the University of Minnesota and Medical Director of MHealth Fairview Point-of-Care Testing; and Christine Wojewoda, MD, FCAP, Director of Clinical Microbiology at the University of Vermont Medical Center, made their case for early vaccination of medical laboratory workers.
“In the laboratory, they are encountering and handling thousands of samples that have active live virus in them,” said Karger, who called clinical laboratory staff and phlebotomists the “forgotten” frontline healthcare workers. “We’re getting 10,000 samples a day. That’s a lot of handling of infectious specimens, and we do want [staff] to be prioritized for vaccination.”
Karger continued to stress the vital role clinical laboratories play not only in COVID-19 testing but also in the functioning of the overall health system. She added that staff burnout is a concern since laboratory staff have been working “full throttle” since March.
“From an operational standpoint, we do need to keep our lab up and running,” she said. “We don’t want to have staff out such that we would have to decrease our testing capacity, which would have widespread impacts for our health system and state.”
Testing for Post-Vaccine Immunity
The CAP panelists also highlighted the need to prepare for the aftermath of widespread COVID-19 vaccinations—the need to test for post-vaccine immunity.
“It’s not routine practice to check antibody levels after getting a vaccine but given the heightened interest in COVID testing, we are anticipating there is going to be some increased in demand for post-vaccine antibody testing,” Karger said. “We’re at least preparing for that and preparing to educate our providers.”
Karger pointed out that clinical pathologists will play an important role in educating providers about the type of antibody tests necessary to test for COVID-19 immunity, because, she says, only the SARS-CoV-2 spike protein antibody test will check for an immune response.
With the pandemic expected to stretch far into 2021, clinical laboratories will continue to play a crucial role in the nation’s healthcare response to COVID-19. As essential workers in the fight against infectious disease, clinical pathologists, clinical chemists, and all medical laboratory staff should be prioritized as frontline healthcare workers.
—Andrea Downing Peck
Related Information:
Pathologists Want First Crack at COVID Vaccines
The Rapidly Changing COVID-19 Testing Landscape
Some Health Care Workers Getting the Vaccine. Other’s Aren’t. Who Decides?
CDC Appendix 2-Terminology: Infection Control in Healthcare Personnel
ASCP Letter: COVID-19 Vaccination Playbook for Jurisdictional Operations
Prioritizing the COVID-19 Vaccine to Protect Patient Access to DiagnosticsCMS Changes Medicare Payment to Support Faster COVID-19 Diagnostic Testing