Half of the genes identified were found to be singletons, unique to specific individuals, offering the possibility of developing precision medicine therapies targeted to specific patients, as well as clinical laboratory tests
Microbiologists and other medical laboratory scientists may soon have more useful biomarkers that aid in earlier, more accurate detection of disease, as well as guiding physicians to select the most effective therapies for specific patients, a key component of Precision Medicine.
Research conducted by scientists from Harvard Medical School and Joslin Diabetes Center into how individual microbial genes in human microbiome may contribute to disease risk uncovered a “staggering microbial gene diversity.”
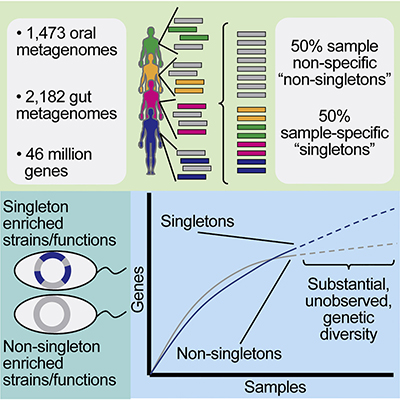
The scientists also found that more than half of the bacterial genes examined occurred only once (called “singletons”) and were specific to each individual. A total of 11.8 million of these singletons came from oral samples and 12.6 million of them derived from gut samples, a Harvard news release noted.
In a paper published in Cell Host and Microbe the researchers state, “Despite substantial interest in the species diversity of the human microbiome and its role in disease, the scale of its genetic diversity, which is fundamental to deciphering human-microbe interactions, has not been quantified.”
To determine this quantity, the researchers conducted a meta-analysis of metagenomes from the human mouth and gut among 3,655 samples from 13 unique studies. Of their findings, they wrote, “We found staggering genetic heterogeneity in the dataset, identifying a total of 45,666,334 non-redundant genes (23,961,508 oral and 22,254,436 gut) at the 95% identity level.”
The scientists also found that while genes commonly found in all the samples seemed to drive the basic functions of a microbe’s survival, the singletons perform more specialized functions within the body, such as creating barriers to protect the micro-organisms from external onslaughts and helping to build up resistance to antibiotics.
“Some of these unique genes appear to be important in solving evolutionary challenges,” said Braden Tierney, a PhD student at Harvard Medical School and one of the authors of the study, in the news release. “If a microbe needs to become resistant to an antibiotic because of exposure to drugs, or suddenly faces a new selective pressure, the singleton genes may be the wellspring of genetic diversity the microbe can pull from to adapt,” he concluded.
‘More Genes in the Human Microbiome than Stars in the Universe’
According to their published paper, the team of microbiologists and bioinformaticians pinpointed more than 46 million bacterial genes contained within 3,655 Deoxyribonucleic acid (DNA) samples. They identified 23,961,508 non-redundant genes in the oral samples and 22,254,436 non-redundant genes in the intestinal samples.
While similar research in the past has targeted bacteria in either the gut or the mouth, the scientists believe their study is the first that analyzed DNA collected from both areas simultaneously.
“Just like no two siblings are genetically identical, no two bacterial strains are genetically identical, either,” said study co-author Chirag Patel, PhD, Assistant Professor of Biomedical Informatics at Harvard’s Blavatnik Institute. “Two members of the same bacterial strain could have markedly different genetic makeup, so information about bacterial species alone could mask critical differences that arise from genetic variation.”
The scientists also endeavored to determine the number of genes that reside in the human microbiome but found the precise number difficult to identify. One calculation estimated that number to be around 232 million, while another suggested the number could be substantially higher.
“Whatever it may be, we hope that our catalog, along with a searchable web application, will have many practical uses and seed many directions of research in the field of host-microbe relationships,” stated Patel in the news release.
New Diagnostics for Clinical Laboratories?
This type of research could have lasting effects on clinical laboratories. As the volume of data generated by diagnostic testing of microbes in patients opens new understanding of how these factors affect human disease and create differences from one individual to another, the increased number of genes and gene mutations mean that microbiology laboratories will increase their use of information technology and analytical software tools.
“Ours is a gateway study, the first step on a what will likely be a long journey toward understanding how differences in gene content drive microbial behavior and modify disease risk,” said Tierney in the Harvard news release.
That’s good news, because new biomarkers derived from such research will help microbiologists and other clinical laboratory scientists more accurately detect disease and identify the best therapies for individual patients.
—JP Schlingman
Related Information:
In a First, Scientists Map the Genetic Diversity of Microbes Residing in the Human Gut and Mouth
The Universe of Microbial Genes
Duke University Study Suggests the Human Body Starves Gut Bacteria to Produce Beneficial Results



