Meaningful Use Stage 3 focuses on interoperability, which is good news for medical laboratories that must spend time and money to develop effective LIS-EHR interfaces
On December 15, 2015, the final rule for Stage 3 meaningful use (MU) went into effect. By now, pathologists and clinical laboratory managers and personnel are well-acquainted with the MU incentive program and the myriad of challenges it presents for almost everyone working in the healthcare sector.
Although the implementation of electronic health records (EHRs) has caused labs some headaches, the Stage 3 MU requirements could reduce some of that pressure. One of the biggest changes in Stage 3, according to the Office of the Federal Register (OFR), is that the ONC is “finalizing changes to remove the menu and core structure of Stage 1 and Stage 2 and reduce the number of objectives to which a provider must attest.” There will be fewer objectives to prove an EHR system is being used in a meaningful way.
That’s good news for providers struggling with EHR attestation. However, the struggle for clinical laboratories isn’t with attestation per se, it’s with interoperability between lab information systems (LIS) and physicians’ EHRs.
Interoperability Is Challenge Medical Labs and Providers
For clinical labs and anatomic pathology groups, lack of interoperability presents a huge, and often costly, barrier. Since labs may work with multiple providers, their LISs must be able to communicate with whatever EHR any given provider uses. If EHRs are more open, and function as platforms rather than closed systems, it is less expensive and generally easier for a lab’s LIS to interface with the various EHRs providers may be using. (See Dark Daily, “Because It Remains Tough to Achieve Interoperability among EHRs, Congress is Proposing Legislation to Resolve That Issue in Ways That May Help Medical Laboratories,” October, 23, 2015.)
Fortunately for labs, the Office of the National Coordinator for Health Information Technology’s (ONC) primary focus in Stage 3 is interoperability. The ONC issued a fact sheet that provides an overview of the final rule. The fact sheet states that the rule “establishes health IT certification criteria that support patient care, patient participation in care delivery, and electronic exchange of interoperable health information.”
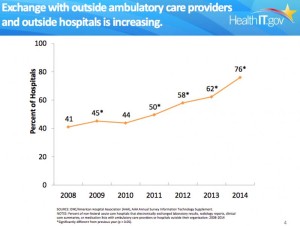
More hospitals are capable of exchanging clinical data with other providers and hospitals. The chart above shows how, beginning with implementation of the Health Information Technology for Economic and Clinical Health (HITECH) Act, passed in 2009, the number of hospitals with this capability has moved from about 45% to 76% at the end of 2014. This is an auspicious development for clinical laboratories, since it is lab test data that makes up the largest proportion of a patient’s health record.
Which Stage 3 Objectives Will Affect Clinical Laboratories
The Stage 3 final rule addresses many areas that will directly affect medical laboratories, including:
- Interoperability: The final rule includes “a Common Clinical Data Set [that is] composed primarily of data expressed using adopted standards and [which is] rigorously testing an identified content exchange standard.”
- Accessibility and Exchange of Data: The final rule “facilitates the accessibility and exchange of data by including enhanced data export, transitions of care, and application programming interface (API) capabilities.”
- Health IT Across the Care Continuum: The final rule “establishes a framework that makes the Office of the National Coordinator (ONC) for Health IT Certification Program open and accessible to more types of health IT, including health IT that supports a variety of care and practice settings, various HHS programs, and public and private interests.”
- Data Segmentation of Sensitive Information: This portion of the final rule “supports the exchange of sensitive health information through the Data Segmentation for Privacy criteria.”
- ONC Dashboard for Reviewing Provider MU Progress: The HealthIT.gov dashboard provides “quick stats” that give some idea of how many hospitals are meeting the Stage 2 MU objectives. Several concern pathology labs. For example, 69.8% of hospitals can “use computerized provider order entry for laboratory tests, radiology tests, and medications.” Other objectives are being more fully met: 88.9% of hospitals have the ability to “incorporate as structured data lab results for more than 40% of patients admitted to inpatient or emergency departments.”
Interoperability versus Information Blocking
EHR companies agree that accomplishing interoperability is a critical step for providers, patients, and third parties such as pathology labs. Stanley Crane, Chief Innovation Officer for Allscripts (NASDAQ:MDRX) in a blog post titled “The Power of Open” stated that “healthcare information technology should be a team sport. When we collaborate with each other, we help each other bring solutions to the healthcare providers and patients.”

Stanley Crane, Chief Innovations Officer, Allscripts, believes that by making the development of Health Information Technology more collaborative “everyone wins: caregivers, healthcare organizations, partners and patients.” (Picture copyright: Allscripts.)
However, a recent article in Politico describes how many EHR vendors included so-called “gag clauses” in their contracts, “effectively forbidding healthcare providers from talking about glitches that slow their work and potentially jeopardize patients,” effectively blocking exchange of possibly critical information between providers. One example, frequently cited by opponents of such clauses, is the prohibition against sharing screenshots without explicitly permission from the EHR vendor.
Elisabeth Belmont, Esq., corporate counsel for MaineHealth, stated in the Politico article that “no EHR software vendor wishes to be ranked by researchers or other third parties as having poor usability or other problematic software issues.” Such restrictions to information exchange could also make it difficult for health information technology (HIT) developers to design LIS-to-EHR interface systems.
The Stage 3 final rule seeks to increase the “reliability and transparency of certified health IT through surveillance and disclosure requirements.” It remains to be seen, however, whether that will extend to making it easier for end users, including the pathologists, scientific staff, and operations team at clinical laboratories, to discuss the specific EHR products with physician-clients.
—Dava Stewart
Related Information:
Tech Firms Refocus To Address Stage 3 Meaningful Use Rules Title
Work Product of the HITPC Meaningful Use Workgroup – Meaningful Use Stage 3 Recommendations
CMS Fact Sheet: EHR Incentive Programs in 2015 and Beyond
Meaningful Use Stage 3 Final Rules Elicit Mixed Reactions
Cancel, Don’t Delay, Meaningful Use Stage 3 for Electronic Health Records



