One device that uses computer algorithms to analyze bodily sounds has undergone clinical trials and is awaiting clearance by the FDA
Is technology poised to render the iconic stethoscope passé? For centuries, doctors have used stethoscopes for listening to functions in different parts of the body. However, new devices now exist that employ electronic biosensors to “listen” in on those same sounds and analyze them with computer algorithms. The devices then deliver assessments to attending physicians.
The concept remains the same. Place a device against the body, listen for sounds, and use those sounds to identify a patient’s symptoms. Since the goal is to monitor sounds, it is not likely that “digital stethoscopes” will impinge on the clinical laboratory testing services provided by pathologists and medical laboratory scientists.
Biosensor More Accurate than Stethoscope, Shows Clinical Trial
One company developing this type of technology is GI Logic of Pasadena, California. Its device is the AbStats, which is designed to help physicians make evidenced-based decisions about which patients can be fed following abdominal surgery.
The biosensor in AbStats is the brainchild of Brennan M.R. Spiegel, M.D., MSHS, Associate Professor of Medicine in the Division of Digestive Diseases, David Geffen School of Medicine at UCLA; the Department of Health Services, UCLA Fielding School of Public Health; and an attending physician in the Division of Gastroenterology, VA Greater Los Angeles Health Care System. Additionally, he is a member of the CURE Digestive Diseases Research Center.
A team of interdisciplinary experts from UCLA’s Wireless Health Institute, under the direction of William J. Kaiser, Ph.D., UCLA Professor of Electrical Engineering and Co-Director of the institute, worked with Dr. Spiegel to develop a disposable, non-invasive acoustic gastro-intestinal surveillance (AGIS) biosensor that is more accurate than the stethoscope for monitoring post-abdominal surgery patients for malfunction in intestinal motility.

Dr. Spiegel believes that his team has invented “a way to monitor a new vital sign—one to go along with heart rate, blood pressure and respiration. This new vital sign, intestinal rate, could prove to be important in diagnosing and treating patients,” he stated in the UCLA press release. (Photo copyright Cedars-Sinai Center for Outcomes Research and Education.)
AbStats Succeeds During Clinical Trials: Awaits FDA Clearance
A clinical trial evaluated the accuracy of the AGIS biosensor technology in detecting patients at risk for Postoperative Ileus (POI), a post-surgical condition that can lead to severe abdominal pain and distention, nausea, vomiting, and other undesirable outcomes. AGIS successfully distinguished POI patients from non-POI patients and healthy controls with 100% sensitivity and 97% specificity, according to the JOGS article.
“After surgery, the bowels shut down under stress as the body is focused on keeping the brain, heart, and lungs alive,” Dr. Spiegel explained. “We also give patients narcotic medications for pain that can cause the bowels to freeze up. The way doctors currently monitor for POI is putting a stethoscope on the patient’s belly for 15 seconds, briefly listening for sounds of intestinal awakening, and asking about flatulence. It’s all very rudimentary and inaccurate,” he continued. “With this new [AGIS] vital sign, the team can now monitor the intestines empirically and make more informed decisions.”
The full results of the UCLA team’s study were published in an October 2014 Journal of Gastrointestinal Surgery (JOGS) article.
The AGIS biosensor serves an unmet need in the field of gastroenterology, stated Dr. Spiegel. AbStats “listens” to the abdomen, detects signals, classifies them, and then provides a measure of the current digestive state. (Video and caption copyright GI Logic.)
AbStats Monitors Bowels Post-Surgery
AbStats, which employs Dr. Spiegel’s AGIS biosensor, is a disposable plastic device embedded with a microphone that attaches to the abdominal wall and connects to a computer that measures acoustic event rates. During a POI event, bowels shut down and the intestinal track becomes quiet, moving just a few times per minute versus 10 times per minute in patients with healthy bowel movement. Bowel movement makes a “clicking” noise that the AGIS biosensor picks up and sends to a computer for analysis, explained a UCLA press release.
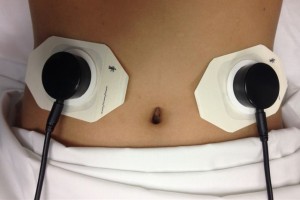
AbStats (pictured above) is an acoustic gastro-intestinal surveillance (AGIS) biosensor device that listens for clicking sounds made when there’s movement in the bowels and then reports them to a connected computer. A computer algorithm uses information from the biosensor to measure acoustic event rates. (Photo copyright UCLA.)
AGIS May be Useful in Diagnosing Other Bowel Ailments
The AGIS biosensor may be useful in diagnosing irritable bowel syndrome and inflammatory bowel disease. There is even the potential for this device to be used in patient self-monitoring. It was suggested that obese patients could be taught how to use AbStats to monitor sounds emanating from their gut to determine when they should or should not eat, which could help them lose weight, pointed out the UCLA press release.
“The role of wearable sensors in healthcare has reached mainstream consciousness and has the capacity to transform how we monitor and deliver care. Yet, there are very few biosensors that are supported by any peer-reviewed evidence,” Dr. Spiegel noted. “This study represents peer-reviewed evidence supporting use of a biosensor; a device born and bred out of UCLA multidisciplinary research.”
Other Digital Devices Also Use Sound to Aid in Diagnosis and Monitoring
AbStats is not the only digital audio device developed to aid physicians at the point of care. Some doctors like Eric Topol, M.D., Director of the Scripps Translational Science Institute in San Diego, have embraced other technologies that replace traditional manual devices. Known as the digital doctor, Topol carries GE’s Vscan, a portable ultrasound device that enables doctors to “see” inside patients during exams.
And in the UK, researchers have developed a portable device that when attached to the surface of the knee identifies and records sounds emanating from within the knee caused by Osteoarthritis, as was reported in an article in The Engineer Magazine.
Implications for Pathologists and Laboratory Managers
These examples are a result of advances in health information technology (HIT). Physicians will continue to use stethoscopes in a traditional manner. Probably more interesting for medical laboratory managers and pathologists is the implication of this research. Improved computer algorithms that can diagnose sounds in the body and deliver actionable information to physicians could create a new channel of information that pathologists might find useful to incorporate into a full clinical pathology consultation in response to a request from an attending physician.
—Patricia Kirk
Related Information:
Validation of an Acoustic Gastrointestinal Surveillance Biosensor for Postoperative Ileu
Abdominal Acoustic Monitoring Can Identify Post-Op Ileus
mHealth Masters: Predicting the End of the Stethoscope




How do I stop people from nerve-wracking me just to make new drugs based on my own relaxing dietary food choices versus my own anxiety levels, that is, wireless heart-rate monitor readings?