Surge of requests for LIS-to-EMR interfaces will soon hit clinical pathology laboratories
EMR adoption by office-based physicians is about to seriously challenge the capability of the nation’s clinical pathology laboratories to quickly build interfaces with the electronic medical record (EMR) systems of their client doctors. That’s the prediction of one of the nation’s foremost experts on how to connect clinical pathology laboratories with physician EMRs.
“Today, only one of four physicians uses electronic medical records. To encourage more physicians to adopt medical record systems over the next five years, the ARRA stimulus package is offering major financial incentives,” observed Pat Wolfram, Vice President of Marketing and Customer Service at Ignis Systems Corporation in Portland, Oregon. “In fact, if the federal government achieves its goals as set forth in the current stimulus package, we can expect more than 300,000 physicians to adopt EMRs over the next five years!”
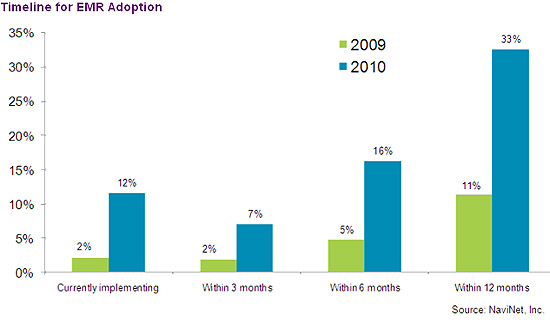
This chart shows the percentage of physician’s who are planning to implement an EHR solution in their practice during the next 12 months in 2009 compared to 2008. It shows a dramatic increase, which reflects the impact of federal financial incentives authorized by the ARRA stimulus Bill. (Source: NaviNet, Inc.)
Wolfram recommends that clinical laboratories and pathology groups track two specific aspects of the intensifying effort to encourage physicians to use EMRs. First is the expectation that tens of thousands of doctors will soon begin the process of adopting an EMR into their medical practice. The second is how federal officials define “meaningful use” of those EMRs and the process used to certify that the physicians use them “meaningfully”.
According to Wolfram, laboratories will soon be inundated with physician requests to interface the lab’s LIS to the physicians’ EMR systems. For example, it required the past 14 years to see 150,000 physicians adopt EMRs. He expects that the rate of physician adoption of EMRs will increase three-fold in the coming year as the only way to meet the federal goal of having 300,000 additional physicians use EMRs during the next five years.
Substantial financial incentives paid by the federal government will be a major driver in this expected wave of physician EMR adoption. But physicians can only collect these bounties if they use the EMR in a “meaningful way”. During the next five years, a total of up to $44,000 in reimbursement from the Centers for Medicare and Medicaid Services can be paid to each doctor who uses an EMR in ways that meet the definition of “meaningful use.”
“If your laboratory’s information technology team is ready to help those physicians make the leap to EMR use, you could earn enormous goodwill and loyalty from the physicians’ practices that you serve,” noted Wolfram. Wolfram says a second element that bears watching is how federal officials define “meaningful use.”
As it stands today, two specific meaningful use criteria apply to how physicians use clinical pathology laboratory tests. “First, care providers will be required to order 80% of their tests, including clinical laboratory tests, within the EMR,” noted Wolfram. “Second, the clinical lab test results must download from the laboratory to automatically populate the patient’s record in the physician’s EMR. At the same time, 50% of those clinical laboratory test results must be ‘structured.’ These structured results allow test result values to be trended, graphed, and drive protocols in the EMR.
“We know from experience that one of the very first features physicians want incorporated into their new EMR system is a direct laboratory results interface to their EMR system,” he added. “Typically, electronic ordering of laboratory tests is a feature that is adopted during a later phase of EMR implementation. However, lab test orders will need to be implemented much earlier and be in active use if that physician wants to satisfy the meaningful use criteria. The recommendation is that your lab should gear up now to help client physicians install electronic ordering of lab tests from within their HER during phase one of the implementation.
“Collectively, these are the reasons why a tsunami of requests from physicians who want your LIS to talk to their EMRs will soon happen,” noted Wolfram. “Unfortunately, many of these EMR products and vendors will be relatively new in the industry and not necessarily experienced in deploying solid lab interfaces. Furthermore, interfaces are very difficult to evaluate and grade when a practice is shopping for an EMR. So the practice may turn to you for advice. Be prepared for that.”
For more on Pat Wolfram’s insights on the HITECH Act and how it will increase physician demand for LIS-to-EMR interfaces, you can check out the audio recording and downloadable materials of a recent audio conference, titled “New Opportunities for Your Laboratory’s LIS-EMR Interface: How to Profit From the “Big Wave” About to Hit Healthcare”, available http://darkdaily.com/audio-conferences
— K. Branz
Related Information:




This is one of the most honest blogs I have ever read. Nothing beats good first hand experience on subjects. Thank you for being honest about this.