Research goal was to isolate circulating tumor cells in venipuncture samples with improved purity compared to standard spiral chips
Many research teams are pursuing the goal of creating assays that detect circulating tumor cells (CTCs) that would allow earlier and more accurate diagnosis of cancer. Now comes news of a unique technology developed at the University of Michigan (U-M) Ann Arbor that showed promised in an early study.
The method of using CTCs to diagnose cancer in patients, while further analyzing specific characteristics of a given cancer case, shows promise as an innovative tool for clinical laboratories and oncologists. However, current approaches face challenges when it comes to proving accuracy and establishing thresholds that might indicate the need for further action.
Researchers at U-M believe they may have solved that problem. They created “Labyrinth,” a “label-free microfluidic device” that condenses 637mm of channels—including 11 loops and 56 corners—onto a 500μm-wide chip that uses inertia and Dean flow to separate white blood cells and CTCs from venipuncture samples at rates as high as 2.5ml per minute. These results improve upon the traditional spiral chip design.
Publishing their findings in Cell Systems, first author of the study Eric Lin, PhD, noted, “With the recent advances in tools for genomic characterization, it is more compelling than ever to look at the tumor heterogeneity to understand tumor progression and resistance to therapies. The Labyrinth device enabled high yields of CTCs without the bias induced by antibody-based selection, allowing the identification of true biological tumor heterogeneity.”
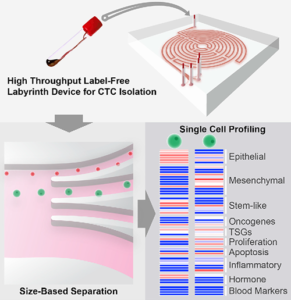
The graphic above, taken from the University of Michigan study, demonstrates the “High-throughput and label-free Labyrinth device that enables single CTC isolation and gene expression characterization.” According to the researchers, “Labyrinth offers a cell-surface marker-independent single-cell isolation platform to study heterogeneous CTC subpopulations.” The U-M study shows promise in creating tools for oncologist and clinical laboratory cancer treatment. (Image copyright: University of Michigan/Cell Systems.)
Challenges in the Isolation of CTCs
The Labyrinth chip is not the first device to assist in isolating CTCs. The U-M study notes that while immune-affinity capture is a validated approach to prognosis, therapeutic monitoring and molecular diagnostics, it does not work with all cancer cases. The researchers also note the method creates challenges in single-cell analysis later.
Existing label-free methods of isolation, such as deterministic lateral displacement, microfluidic flow fractionation, and acoustic-based separation, avoid these concerns but face issues of their own. The researchers noted, “Issues encountered with these approaches include pore clogging, high-pressure drop, pre-fixation to prevent CTC loss, low throughput, and excessive non-specific cell retention.”
The researchers further clarified that a major factor separating the Labyrinth chip from other methods is the ability to identify CTC subpopulations without the need for manual selection based on positive or negative protein expression. Thus, improving the ability to conduct further single-cell analysis from the results. Testing of the Labyrinth chip involved a variety of cancer cell lines, including:
· Human breast (MCF-7);
· Pancreatic (PANC-1);
· Prostate (PC-3); and,
· Lung (H1650).
And while standard spiral chips are already a common method for conducting size-based sorting, the purity of results is less than ideal with thousands of other cells remaining in the sample.
The researchers reported that the Labyrinth chip recovered 91.5% (plus or minus 0.9%) of cancer cells and removed 91.4% (plus or minus 3.3%) of white blood cells in a spiked buffer test.
“Bigger cells, like most cancer cells, focus pretty fast due to the curvature. But the smaller the cell is, the longer it takes to get focused,” Sunitha Nagrath, PhD, Associate Professor of Chemical Engineering and a lead developer of the Labyrinth chip, stated in a U-M news release. “The corners produce a mixing action that makes the smaller white blood cells come close to the equilibrium position much faster.”
Labyrinth also supports a series configuration of multiple chips. While testing two chips in series, researchers noted “a two-log improvement in tumor cell enrichment over the single Labyrinth.” They claim this is a higher purity than other label-free methods they studied, while adding only five minutes to processing times.

Sunitha Nagrath, PhD (above), is an Associate Professor of Chemical Engineering at the University of Michigan, and one of the lead developers of the Labyrinth chip. “You cannot put a box around these cells,” she noted in the U-M news release. “The markers for them are so complex, there is no one marker we could target for all these stages.” (Photo copyright: University of Michigan.)
Current Testing Using the Labyrinth Chip
The chip is already in use in a clinical trial for an aggressive form of breast cancer by Max Wicha, MD, Madeline and Sidney Forbes Professor of Oncology, Founding Director Emeritus, University of Michigan Comprehensive Cancer Center, and co-author of the Cell Systems study, who lead the study along with Nagrath.
The trial involves the attempted activation of adult system cells by blocking the signaling molecule interleukin-6. Wicha suspects the molecule enables cancer stem cells as well. “We think that this may be a way to monitor patients in clinical trials,” he said in the U-M news release. “Rather than just counting the cells, by capturing them, we can perform molecular analysis [to] know what we can target with treatments.”
The news release further highlights how this chip is specifically suited to such a task. As cancer stem cells transition from stem-like cells to more ordinary cell types, their gene expression shifts as well. This creates an issue when using conventional cell targeting. Nagrath notes this concern, stating, “The markers for [cancer stem cells] are so complex, there is no one marker we could target for all these stages.”
The Labyrinth chip shows potential for overcoming one of the biggest hurdles to leveraging CTCs to diagnose cancers and develop personalized therapies. Currently, the chip can output to Fluidigm, DEPArray by Silicon Biosystems, and RainDance Technologies’ RainDrop Digital PCR System.
The U-M researchers hope that future research will yield additional applications and compatible systems to further improve the ability for medical laboratories to use CTCs in the early detection and monitoring of cancer cases.
—Jon Stone
Related Information:
‘Labyrinth’ Chip Could Help Monitor Aggressive Cancer Stem Cells
High-throughput Microfluidic Labyrinth for the Label-free Isolation of Circulating Tumor Cells
Novel Labyrinth Chip Monitors Cancer Stem Cells in Clinical Trial
‘Labyrinth’ Device Sorts Cancer Cells from Healthy Blood
This Awesome Blood Labyrinth Is the Newest Method for Catching Cancer Cells
Labyrinth Chip Has the Potential to Lead to Customized Cancer Treatments


