Oct 22, 2018 | Digital Pathology, Instruments & Equipment, Laboratory Instruments & Laboratory Equipment, Laboratory Management and Operations, Laboratory News, Laboratory Pathology, Laboratory Testing
UK study shows how LDTs may one day enable physicians to identify patients genetically predisposed to chronic disease and prescribe lifestyle changes before medical treatment becomes necessary
Could genetic predisposition lead to clinical laboratory-developed tests (LDTs) that enable physicians to assess patients’ risk for specific diseases years ahead of onset of symptoms? Could these LDTs inform treatment/lifestyle changes to help reduce the chance of contracting the disease?
A UK study into the genetics of one million people with high blood pressure reveals such tests could one day exist.
Researchers at Queen Mary University of London and Imperial College London uncovered 535 new gene regions affecting hypertension in the largest ever worldwide genetic study of blood pressure, according to a news release.
They also confirmed 274 loci (gene locations) and replicated 92 loci for the first time.
“This is the most major advance in blood pressure genetics to date. We now know that there are over 1,000 genetic signals which influence our blood pressure. This provides us with many new insights into how our bodies regulate blood pressure and has revealed several new opportunities for future drug development,” said Mark Caulfield, MD,
Professor of Clinical Pharmacology at Queen Mary University of London, in the news release. He is also Director of the National Institute for Health Research Barts Biomedical Research Centre.
The researchers believe “this means almost a third of the estimated heritability for blood pressure is now explained,” the news release noted.
Clinical Laboratories May Eventually Get a Genetic Test Panel for Hypertension
Of course, more research is needed. But the study suggests a genetic test panel for hypertension may be in the future for anatomic pathologists and medical laboratories. Physicians might one day be able to determine their patients’ risks for high blood pressure years in advance and advise treatment and lifestyle changes to avert medical problems.
By involving more than one million people, the study also demonstrates how ever-growing pools of data will be used in research to develop new diagnostic assays.
The researchers published their study in Nature Genetics.

The video above summarizes research led by Queen Mary University of London and Imperial College London, which found over 500 new gene regions that influence people’s blood pressure, in the largest global genetic study of blood pressure to date. Click here to view the video. (Photo and caption copyright: Queen Mary University of London.)
Genetics Influence Blood Pressure More Than Previously Thought
In addition to identifying hundreds of new genetic regions influencing blood pressure, the researchers compared people with the highest genetic risk of high blood pressure to those in the low risk group. Based on this comparison, the researchers determined that all genetic variants were associated with:
- “having around a 13 mm Hg higher blood pressure;
- “having 3.34 times the odds for increased risk of hypertension; and,
- “1.52 times the odds for increased risk of poor cardiovascular outcomes.”
“We identify 535 novel blood pressure loci that not only offer new biological insights into blood pressure regulation, but also highlight shared genetic architecture between blood pressure and lifestyle exposures. Our findings identify new biological pathways for blood pressure regulation with potential for improved cardiovascular disease prevention in the future,” the researchers wrote in Nature Genetics.
Other Findings Link Known Genes and Drugs to Hypertension
The UK researchers also revealed the Apolipoprotein E (ApoE) gene’s relation to hypertension. This gene has been associated with both Alzheimer’s and coronary artery diseases, noted Lab Roots. The study also found that Canagliflozin, a drug used in type 2 diabetes treatment, could be repurposed to also address hypertension.
“Identifying genetic signals will increasingly help us to split patients into groups based on their risk of disease,” Paul Elliott, PhD, Professor, Imperial College London Faculty of Medicine, School of Public Health, and co-lead author, stated in the news release. “By identifying those patients who have the greatest underlying risk, we may be able to help them to change lifestyle factors which make them more likely to develop disease, as well as enabling doctors to provide them with targeted treatments earlier.”
Working to Advance Precision Medicine
The study shares new and important information about how genetics may influence blood pressure. By acquiring data from more than one million people, the UK researchers also may be setting a new expectation for research about diagnostic tests that could become part of the test menu at clinical laboratories throughout the world. The work could help physicians and patients understand risk of high blood pressure and how precision medicine and lifestyle changes can possibly work to prevent heart attacks and strokes among people worldwide.
—Donna Marie Pocius
Related Information:
Study of One Million People Leads to World’s Biggest Advance in Blood Pressure Genetics
Researchers Find 535 New Gene Regions That Influence Blood Pressure
Genetic Analysis of Over One Million Identifies 535 New Loci Associated with Blood Pressure Traits
The Facts About High Blood Pressure
High Blood Pressure Breakthrough: Over 500 Genes Uncovered
Study of a Million People Reveals Hypertension Genes
Oct 8, 2018 | Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing
Both health systems will use their EHRs to track genetic testing data and plan to bring genetic data to primary care physicians
Clinical laboratories and pathology groups face a big challenge in how to get appropriate genetic and molecular data into electronic health record (EHR) systems in ways that are helpful for physicians. Precision medicine faces many barriers and this is one of the biggest. Aside from the sheer enormity of the data, there’s the question of making it useful and accessible for patient care. Thus, when two major healthcare systems resolve to accomplish this with their EHRs, laboratory managers and pathologists should take notice.
NorthShore University HealthSystem in Illinois and Geisinger Health System in Pennsylvania and New Jersey are working to make genetic testing part of primary care. And both reached similar conclusions regarding the best way for primary care physicians to make use of the information.
One area of common interest is pharmacogenomics.
At NorthShore, two genetic testing programs—MedClueRx and the Genetic and Wellness Assessment—provide doctors with more information about how their patients metabolize certain drugs and whether or not their medical and family histories suggest they need further, more specific genetic testing.
“We’re not trying to make all of our primary care physicians into genomic experts. That is a difficult strategy that really isn’t scalable. But we’re giving them enough tools to help them feel comfortable,” Peter Hulick, MD, Director of the Center for Personalized Medicine at NorthShore, told Healthcare IT News.
Conversely, Geisinger has made genomic testing an automated part of primary care. When patients visit their primary care physicians, they are asked to sign a release and undergo whole genome sequencing. An article in For the Record describes Geisinger’s program:
“The American College of Medical Genetics and Genomics classifies 59 genes as clinically actionable, with an additional 21 others recommended by Geisinger. If a pathogenic or likely pathogenic variant is found in one of those 80 genes, the patient and the primary care provider are notified.”

William Andrew Faucett (left) is Director of Policy and Education, Office of the Chief Scientific Officer at Geisinger Health; and Peter Hulick, MD (right), is Director of the Center for Personalized Medicine at NorthShore University HealthSystem. Both are leading programs at their respective healthcare networks to improve precision medicine and primary care by including genetic testing data and accessibility to it in their patients’ EHRs. (Photo copyrights: Geisinger/NorthShore University HealthSystem.)
The EHR as the Way to Access Genetic Test Results
Both NorthShore and Geisinger selected their EHRs for making important genetic information accessible to primary care physicians, as well as an avenue for tracking that information over time.
Hulick told Healthcare IT News that NorthShore decided to make small changes to their existing Epic EHR that would enable seemingly simple but actually complex actions to take place. For example, tracking the results of a genetic test within the EHR. According to Hulick, making the genetic test results trackable creates a “variant repository,” also known as a Clinical Data Repository.
“Once you have that, you can start to link it to other information that’s known about the patient: family history status, etc.,” he explained. “And you can start to build an infrastructure around it and use some of the tools for clinical decision support that are used in other areas: drug/drug interactions, reminders for flu vaccinations, and you can start to build on those decision support tools but apply them to genomics.”
Like NorthShore, Geisinger is also using its EHR to make genetic testing information available to primary care physician when a problem variant is identified. They use EHR products from both Epic and Cerner and are working with both companies to streamline and simplify the processes related to genetic testing. When a potentially problematic variant is found, it is listed in the EHR’s problem list, similar to other health issues.
Geisinger has developed a reporting system called GenomeCOMPASS, which notifies patients of their results and provides related information. It also enables patients to connect with a geneticist. GenomeCOMPASS has a physician-facing side where primary care doctors receive the results and have access to more information.
Andrew Faucett, Senior Investigator (Professor) and Director of Policy and Education, Office of the Chief Scientific Officer at Geisinger, compares the interpretation of genetic testing to any other kind of medical testing. “If a patient gets an MRI, the primary care physicians doesn’t interpret it—the radiologist does,” adding, “Doctors want to help patients follow the recommendations of the experts,” he told For the Record.
The Unknown Factor
Even though researchers regularly make new discoveries in genomics, physicians practicing today have had little, if any, training on how to incorporate genetics into their patients’ care. Combine that lack of knowledge and training with the current lack of EHR interoperability and the challenges in using genetic testing for precision medicine multiply to a staggering degree.
One thing that is certain: the scientific community will continue to gather knowledge that can be applied to improving the health of patients. Medical pathology laboratories will play a critical role in both testing and helping ensure results are useful and accessible, now and in the future.
—Dava Stewart
Related Information:
Introducing “Genomics and Precision Health”
How NorthShore Tweaked Its Epic EHR to Put Precision Medicine into Routine Clinical Workflows
Precise, Purposeful Health Care
Next-Generation Laboratory Information Management Systems Will Deliver Medical Laboratory Test Results and Patient Data to Point of Care, Improving Outcomes, Efficiency, and Revenue
Aug 24, 2018 | Instruments & Equipment, Laboratory Instruments & Laboratory Equipment, Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing
CDC reports more than 93-million US adults are obese, and health issues related to obesity include heart disease, stroke, type 2 diabetes, and cancers
In recent years, the role of the human microbiome in weight loss or weight gain has been studied by different research groups. There is keen interest in this subject because of the high rates of obesity, and diagnostic companies know that development of a clinical laboratory test that could assess how an individual’s microbiome affects his/her weight would be a high-demand test.
This is true of a study published this year in Mayo Clinic Proceedings. Researchers at Mayo Clinic looked at obese patients who were in an active lifestyle intervention program designed to help them lose weight. It was determined that gut microbiota can have a role in both hindering weight loss and supporting weight loss.
Gut Microbiota More Complicated than Previously Thought
The Mayo researchers determined “an increased abundance of Phascolarctobacterium was associated with [successful weight loss]. In contrast, an increased abundance of Dialister and of genes encoding gut microbial carbohydrate-active enzymes was associated with failure to [lose] body weight. A gut microbiota with increased capability for carbohydrate metabolism appears to be associated with decreased weight loss in overweight and obese patients undergoing a lifestyle intervention program.”
How do bacteria impede weight loss? Vandana Nehra, MD, Mayo Clinic Gastroenterologist and co-senior author of the study, explained in a news release.
“Gut bacteria have the capacity to break down complex food particles, which provides us with additional energy. And this is normally is good for us,” she says. “However, for some individuals trying to lose weight, this process may become a hindrance.”
Put another away: people who more effectively metabolized carbohydrates were the ones who struggled to drop the pounds, New Atlas pointed out.

Vandana Nehra, MD (left), and Purna Kashyap, MBBS (right), are Mayo Clinic Gastroenterologists and co-senior authors of the Mayo study. “While we need to replicate these findings in a bigger study, we now have an important direction to pursue in terms of potentially providing more individualized strategies for people who struggle with obesity,” Nehra noted in the news release. Thus, precision medicine therapy for obese individuals could be based on Mayo Clinic’s research. (Photo copyright: Mayo Clinic.)
Mayo Study Provides Clues to Microbiota Potential in Weight Loss
The Mayo researchers wanted to know how gut bacteria behave in people who are trying to lose weight.
They recruited 26 people, ranging in age from 18 to 65, from the Mayo Clinic Obesity Treatment Research Program. Fecal stool samples, for researchers’ analysis, were collected from participants at the start of the three-month study period and at the end. The definition of successful weight loss was at least 5% of body weight.
Researchers found the following, according Live Science:
- 2 lbs. lost, on average, among all participants;
- Nine people were successful, losing an average of 17.4 lbs.;
- 17 people did not meet the goal, losing on average just 3.3 lbs.; and,
- More gut bacterial genes that break down carbohydrates were found in stool samples of the unsuccessful weight loss group, as compared to the successful dieters.
The researchers concluded that “An increased abundance of microbial genes encoding carbohydrate-active enzyme pathways and a decreased abundance of Phascolarctobacterium in the gut microbiota of obese and overweight individuals are associated with failure to lose at least 5% weight following a 3-month comprehensive lifestyle intervention program.”
Purna Kashyap, MBBS, Mayo Clinic Gastroenterologist and co-senior author of the study, told Live Science, “The study suggests there is a need to take the microbiome into account in clinical studies (on weight loss), and it also provides an important direction to pursue in terms of providing individualized care in obesity.” The very basis of precision medicine.
Future Weight-Loss Plans Based on Patient’s Microbiota
The Mayo Clinic researchers acknowledged the small sample size and need for more studies with larger samples over a longer time period. They also noted in their paper that Dialister has been associated with oral infections, such as gingivitis, and its role in energy expenditure and metabolism is unclear.
Still, the study suggests that it may soon be possible to give people individualized weight loss plans based on their gut bacteria. Clinical laboratory professionals and pathologists will want to stay abreast of follow-up studies and replication of findings by other research teams. A future medical laboratory test to analyze patients’ microbiomes could help obese people worldwide as well as lab business volume.
—Donna Marie Pocius
Related Information:
Gut Microbial Carbohydrate Metabolism Hinders Weight Loss in Overweight Adults Undergoing Lifestyle Intervention with a Volumetric Diet
Gut Microbiota from Twins Discordant for Obesity Modulate Metabolism in Mice
CDC: Adult Obesity Facts
Makeup of an Individual’s Gut Bacteria May Play Role in Weight Loss, Mayo Study Suggests
Struggle to Lose Weight? Your gut Bacteria May Be to Blame
Your Gut Bacteria May Make It Harder to Lose Weight
Diet Hit a Snag? Your Gut Bacteria May be Partly to Blame
Can’t Lose Weight? Your Gut Bacteria Could be to Blame, According to Study
Richness of Human Gut Microbiome Correlates with Metabolic Markers
Annual Medical Spending Attributable to Obesity: Payer- and Service-Specific Estimates
5 Ways Gut Bacteria Affect Your Health
Cornell Researchers Identify Gut Microbes That May Help Some People Remain Thin; Findings Could Result in Clinical Laboratory Tests to Analyze Microbiomes of Individuals
Clinical Laboratories Might Soon be Diagnosing Obesity and Guiding Therapies that Utilize Engineered Microbes
Aug 23, 2018 | Digital Pathology, Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing, Management & Operations
Genetic testing, gene sequencing done by clinical laboratories and anatomic pathology groups underpin how first-mover hospitals, health networks are improving patient outcomes
In just a few weeks, an unprecedented gathering will bring together the nation’s most prominent first-mover health networks, hospitals, and companies operating programs that deliver precision medicine daily to patients in clinical care settings.
On Sept. 12-13, 2018, “Breakthroughs with Genetic and Precision Medicine: What All Health Network CEOs Need to Know,” will take place at the Hutton Hotel in Nashville, Tenn. “What differentiates these sessions is the emphasis on each organization’s strategy, how it launched its precision medicine programs, what is improving in patient outcomes, and how payers are reimbursing for these services,” stated Robert L. Michel, Executive Director of the Precision Medicine Institute in Austin, Texas. “This is not about the science of precision medicine. Rather, it is about the practical elements required for any hospital, health system, or physician group to actually set up and deliver a precision medicine service to patients on a daily basis.”
Precision Medicine’s First-Mover Hospitals and Providers to Speak
Health systems and hospitals headlining this special conference are:
Companies scheduled to present include:
- Illumina;
- Humana;
- Sonic Healthcare USA;
- Genome Medical;
- CQuentia, and,
- S. HealthTek.
Exhibitors include the above, plus: Thermo Fisher, Philips, Sunquest, and MyGenetx.
“This meeting will give you the insider’s understanding about delivering precision medicine in real patient care settings that cannot be accessed at other venues,” noted Michel. “The goal is to have first-mover providers share their experiences, thus providing a road map that other hospitals, physician practices, and other providers at this conference can take back and follow with confidence.”
Michel said that sessions will be dedicated to precision medicine strategies, how it is being used in oncology, primary care, the role of pharmacogenomics, and use of healthcare big data. Speakers will describe the clever ways innovative health networks and hospitals are using healthcare big data to inform physicians in ways that improve outcomes, lower the cost of care and, in two real-world case studies, are generating seven-figure reimbursement from shared savings programs with certain health plans.

This year’s keynote address is by Jeffrey R. Balser, MD, PhD (above), President and CEO, Vanderbilt University Medical Center and Dean of the Vanderbilt University School of Medicine, one of the most progressive and innovative health systems in the country. (Photo copyright: Vanderbilt University.)
Using Healthcare Big Data to Achieve Precision Medicine Success, Shared Savings
“Shared savings successes will be one of the breakthrough achievements reported at the Nashville event,” he explained. “We’ve invited two prominent provider organizations to share how they are using healthcare big data to support physicians in achieving improved patient outcomes while at the same time impressively reducing the overall cost of care. To my knowledge, this is the first time these precision medicine case studies have been presented at a national meeting.”
One such presentation will be delivered by Philip Chen, MD, PhD, Chief Healthcare Informatics Officer at Sonic Healthcare USA Austin, Texas. Their precision medicine goal was to use healthcare big data to help physicians better manage diabetes and other chronic conditions in their practices. This program involved a large primary care practice and a major health insurer. Now in its fourth year, Sonic Healthcare USA is earning six- and seven-figure payments as part of a shared savings arrangement with the insurer.
“Shared savings is definitely a Holy Grail for all large health networks and physician groups as payers drop fee-for-service and switch providers to value-based payments,” said Michel. “The experience of Sonic Healthcare in this innovative three-way collaboration with an insurer and a very large physician group demonstrates that a strong data analytics capability and engagement with physicians can simultaneously bend the cost-of-care-curve downward while improving patient outcomes, as measured year-by-year. This is a presentation every C-Suite executive should attend.
Strategic, Business, Operational, and Financial Aspects of Precision Medicine
“This conference—centered upon the strategic, business, operational, and financial aspects of a precision medicine program—came to be because it is the unmet need of every health network CEO and C-Suite administrator,” observed Michel. “Every healthcare leader tasked with developing an effective clinical and financial strategy for his or her institution knows that the real challenge in launching a precision medicine program for patient care is not the science.
“Rather, the true challenges come from how to support clinical needs with the availability of capital, recruiting experienced clinicians, and putting the right informatics capabilities in place,” he stated. “Most hospital and health network administrators recognize the risk of launching a precision medicine program too early. They know such programs can suck up huge amounts of resources without producing significant improvements in patient care. What adds to the risk is that payers may be slow to reimburse for precision medicine.”
Register today to guarantee your place at “Breakthroughs with Genetic and Precision Medicine: What All Health Network CEOs Need to Know,” (or copy and paste this URL in your browser: https://dark.regfox.com/precision-medicine-institute).
Register by September 1 and save $300 on tuition! Plus, take advantage of our special Team Discount Program, so you and your key team members can get the most out of the conference by attending together.
“Breakthroughs with Genetic and Precision Medicine: What All Health Network CEOs Need to Know” is the gold-standard summit for everyone active or interested in succeeding with precision medicine programs. Don’t miss out—register today!
—Michael McBride
Related Information:
Breakthroughs with Genetic and Precision Medicine: What All Health Network CEOs Need to Know—Full Agenda and Details
Breakthroughs with Genetic and Precision Medicine: What All Health Network CEOs Need to Know—Registration information
Ongoing Growth in Consumer Genetic Testing Pressures Hospitals, Healthcare Networks to Educate and Prepare Physicians
Syapse Creates Precision Medicine Council That Quickly Attracted 200 of the Biggest Hospitals and Health Networks as Members
When Ramping Up Genomic Programs, Health Network/Hospital CEOs and Executives Must Consider Emerging Technologies, Swiftly Rising Consumer Demand
Precision Medicine Success Hinges on Diagnostics’ Clinical Utility
Precision Medicine and Sharing Medical Data in Real Time: Opportunities and Barriers
Ongoing Growth in Volume of Clinical Laboratory Tests That Support Precision Medicine Due to Physician Acceptance; Payers Still Have Concerns
Aug 20, 2018 | Instruments & Equipment, Laboratory Instruments & Laboratory Equipment, Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing, Management & Operations
“On-a-chip” devices continue to advance and medical laboratories will be natural repositories for patient data as the technology continues to improve
Dark Daily has predicted that the future of clinical laboratory testing will include highly complex multi-analyte test panels. The biomarkers, however, could number in the hundreds or thousands. So, it’s interesting to see new research by a Massachusetts Institute of Technology (MIT) team currently developing a multi-biomarker organ test device for clinical purposes.
Motivated by the costly failure of animal testing efforts to develop drug safety and efficacy in humans, the MIT research engineers created a microfluidic platform technology they dubbed “physiome-on-a-chip,” or more colloquially, “body-on-a-chip.” Their goal is to identify drug reaction in different cell groups within the body (in vivo).
They acknowledged contributions of in vitro microphysiological systems (MPSs), AKA “organ-on-a-chip” (OOC) systems. They note, however, in their paper published in Scientific Reports, that more complex systems that interconnect and receive data from multiple MPSs are needed due to increasing limitations arising from drugs’ “lack of efficacy” rather than toxicity.
“Here we describe the development and implementation of multi-MPS platforms, AKA physiome-on-a-chip, supporting four-way, seven-way, and 10-way MPS interactions for several weeks,” the MIT engineers wrote.
Though MIT’s new technology needs further research and development time, as well as clinical trials, this type of chip design and its ability to scale is a positive development and progress toward Dark Daily’s prediction. Once finalized, it could be adopted in medical laboratories for many types of diagnostic testing purposes.
Researchers Motivated to Improve Drug Efficacy
According to an MIT news release, “MIT engineers have developed new technology that could be used to evaluate new drugs and detect possible side effects before the drugs are tested in humans. Using a microfluidic platform that connects engineered tissues from up to 10 organs, the researchers can accurately replicate human organ interactions for weeks at a time, allowing them to measure the effects of drugs on different parts of the body.”
The “body-on-a-chip” technology, MIT says, is aimed at determining how drugs may affect one organ while also having side effects on others.
“Some of these effects are really hard to predict from animal models because the situations that lead to them are idiosyncratic. With our chip, you can distribute a drug and then look for the effects on other tissues and measure the exposure and how it is metabolized,” said Linda Griffith, PhD, Professor of Teaching Innovation at MIT’s School of Engineering, and a senior author of the study, in the news release.
According to MIT, factors affecting the effectiveness of pharmaceuticals may include:
- Genetics;
- Environment;
- Personal lifestyles; and,
- Interactions with other drugs.
TechCrunch called the study “unprecedented,” pointing to the platform’s connection of so many tissues and the technology’s ability to keep them stable for weeks.

“An advantage of our platform is that we can scale it up or down and accommodate a lot of different configurations,” Linda Griffith, PhD, MIT Professor, MIT School of Engineering, told Science Daily. “I think the field is going to go through a transition where we start to get more information out of a three-organ or four-organ system, and it will start to become cost-competitive because the information you’re getting is so much more valuable.” (Photo copyright: MacArthur Foundation.)
How “Body-on-a-Chip” Works
“Body-on-a-chip” is about the size of a tablet computer and links 10 organ types, including: liver, lung, gut, endometrium, brain, heart, pancreas, kidney, skin, and skeletal muscle.
Using microfluidic platform technology, the researchers placed one- to two-million cells from human tissue samples into the device and then pushed fluid through the chip to resemble blood flow, the Daily Mail reported, adding that MIT’s MPS platform design features:
- Compartments made from a plastic block;
- Passages for fluid to move (as a circulatory system does) between the compartments;
- A water reservoir to limit fluid evaporation; and,
- Ability to monitor flow of molecular exchanges and drug distribution.
Essentially, using the MIT device, a drug can be introduced to one organ, processed normally, and then passed to other organs for processing and use in other ways, TechCrunch summarized.
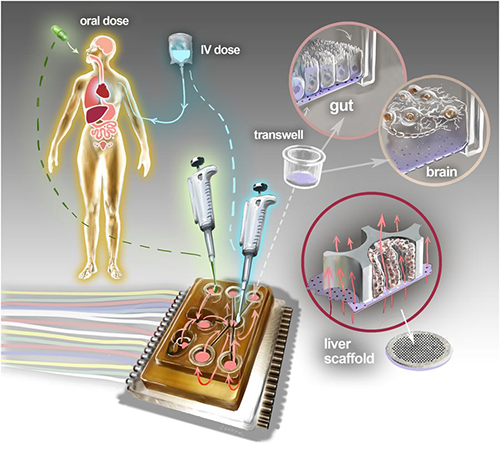
The physiome-on-a-chip system (above schematic) comprises bioengineered devices that nurture many interconnected 3D MPSs representing specified functional behaviors of each organ of interest, designed to capture essential features of in vivo physiology based on quantitative systems models tailored for individual applications such as drug fate or disease modeling. This technology could eventually be utilized for clinical laboratory and anatomic pathology testing. (Image and caption copyright: Victor O. Leshyk/Scientific Reports.)
Drug Delivery, Effects on Multiple Tissues Noted in MIT Study
The MIT researcher engineers reported these findings and accomplishments:
- Delivering a drug to the gastrointestinal tissue;
- Replicating digesting a drug;
- Observing as a drug was transported to other tissues and metabolized;
- Measuring a drug’s path; and,
- Noting effects of a drug on different tissues and how drugs break down.
“The huge potential of MPS technology is revealed by connecting multiple organ chips in an integrated system for in vitro pharmacology. This study beautifully illustrates that multi-MPS ‘physiome-on-a-chip’ approaches, which combine the genetic background of human cells with physiologically relevant tissue-to-media volumes, allow accurate prediction of drug pharmacokinetics and drug absorption, distribution, metabolism, and excretion,” said Kevin Healy, PhD, Professor of Bioengineering and Materials Science and Engineering, at University of California Berkeley in the MIT news release. Healy was not involved in the research.
Unique Device Design
In addition to making it possible to study so many different tissue types, the device design, according to MIT, is unique for these reasons:
- Its open microfluidic system, rather than a closed system, means the lid can be removed to manipulate tissue samples;
- Instead of external pumps common in closed systems, the MIT team used “on-board pumps” to control flow of liquid between the organs; and,
- The pumps used enabled larger engineered tissues, such as those from tumors in an organ, to be assessed.
The MIT engineers next plan to focus on specific organs—including the brain, liver, and gastrointestinal tissue—to model Parkinson’s disease, Digital Trends reported.
As healthcare providers and medical laboratories adopt precision medicine, MIT’s contributions are both timely and important. The ability to accommodate many different configurations in one platform is impressive, and something Dark Daily has been anticipating.
—Donna Marie Pocius
Related Information:
A “Body-on-a-Chip” Strings Together 10 Model Human Organs
“Body-on-a-Chip” Could Improve Drug Evaluation
MIT Builds “Body-on-a-Chip” Device That Can Store up to 10 Artificial Organs at Once
Interconnected Microphysiological Systems for Quantitative Biology and Pharmacology Studies
MIT Gadget Puts Multiple Artificial Organs into a Paperback-Sized Connected System
Drug Testing Could Get a Boost from MIT’s “Body-on-a-Chip”








